Welcome to Where's My Flying Car? (WMFC), a Substack about the business of science.
We'll shine a spotlight on the latest scientific breakthroughs and the people behind the breakthrough, problems in scientific business processes, practical solutions and how we can all be a part of shaping the future.
Articles you can read in less than 5 minutes to bridge the knowledge and communication gap between scientists, IT, and finance.
WMFC For People in a Hurry
Key Takeaways:
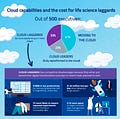
The Society for Laboratory Automation and Screening (SLAS) San Diego’s conference attendance increased more than 40% from 2022 to 2023, with over 7300 researchers, scientists, data engineers, data scientists, and informatics professionals in attendance this year.
As per the survey of CFOs by PwC, more than 68% of CFOs planned to focus their investment efforts on digital transformation with automation as a priority.
Only 50% of scientific data is sufficiently prepared and available for analysis and data science, setting data science teams up for failure, and causing a mass transition of data scientists forced to step into data engineering roles.
Purpose-built vendor-agnostic scientific data platforms are in growing demand to streamline workflows and centralize data for improved efficiency and accuracy in experiments.
A collaborative network of vendors are needed to provide integrated solutions for mutual customers that can easily integrate with existing instrumentation and software.
Cross-domain expertise is crucial in guiding companies throughout the scientific data journey.
Misconceptions About AI & Automation
The Society for Laboratory Automation and Screening (SLAS) is a powerhouse of innovation, collaboration, and excellence in scientific research. With its international conferences, symposia, and publications, SLAS leads the way in promoting the pursuit of scientific excellence and advancing the field of laboratory automation.
At the 2023 SLAS conference in San Diego, attendees from across the academic, industry, and government sectors came together to share their insights, research, and innovations in laboratory automation. From the latest advances in artificial intelligence and machine learning to the development of new techniques and tools for data analysis, the conference was a treasure trove of insights and knowledge for anyone interested in advancing scientific research.
Despite the tremendous advancements in robotics and software, innovation seems to occur in isolation. Automating parts of discovery, development, and manufacturing; but barely scratching the surface of true connectivity across the drug development lifecycle.
The true barrier to achieving the same level of automation realized in automobile manufacturing?
Data standardization and interoperability.
Biopharma companies all share the same noble goal - bring better safer therapeutics to patients faster and improve human health at affordable prices.
But the current reality is grim - it takes a staggering 10 years and costs an average of $1.1 billion USD, with a total cost of up to $4.54 billion for each newly approved drug. This is unacceptable. We need to find a way to accelerate drug discovery, development, and delivery.
Biopharmas have recognized the power of technologies like AI and ML to build predictive models from existing data sets. But there's an all too common oversight - AI/ML outcomes heavily rely on the quality of underlying data.
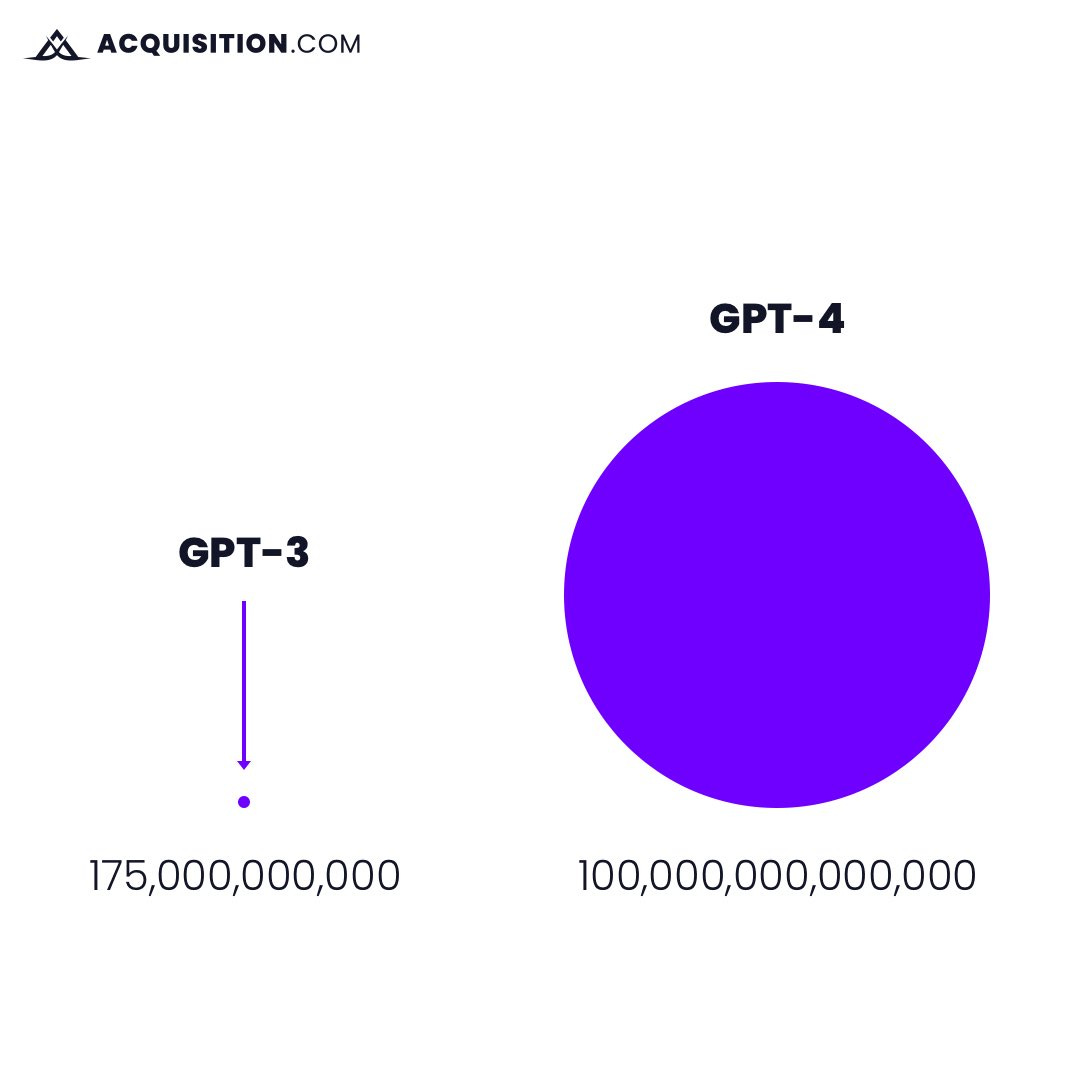
Let’s take ChatGPT for example, who gained one million users just five days after launching in November of last year and created a frenzy of interest for generative AI.
Why does it work so well?
The answer is found in their intelligently engineered underlying dataset
In the visual below, the first dot represents the amount of data ChatGPT 3 was trained on. The second is what ChatGPT 4 is currently being trained on.
You don’t have to look very hard to find this sort of example in biopharma, but they are far and few between.
DeepMind's AlphaFold is a deep learning-based system that predicts the 3D structures of proteins. Proteins are fundamental building blocks of life and understanding their structures can help scientists better understand their functions and develop new drugs. AlphaFold uses deep neural networks to predict protein structures with remarkable accuracy, which was previously considered a major challenge in the field of protein folding.
The underlying training data set is extremely important in AlphaFold's ability to make accurate predictions of protein structures. AlphaFold was trained on a vast amount of data, including the known structures of proteins, as well as protein sequences and other data. The training data set needed to be large and diverse enough to allow AlphaFold to learn the complex patterns and relationships that exist between protein sequences and their corresponding structures.
Without high-quality training data, AlphaFold would not have been able to make accurate predictions of protein structures. The quality and quantity of the training data are critical to the accuracy of the system, as AlphaFold relies on patterns in the data to make predictions. Additionally, the accuracy of the training data is important, as errors or inaccuracies in the data can lead to incorrect predictions.
So, why is FAIR scientific data so important in biopharma? Because it impacts three critical industry drivers:
First, speed - we need to learn from experiments faster and reduce time to market for final molecules and biologics.
Second, cost - we must improve productivity, increase operational efficiency, and get better returns on investment.
And third, risk - we need to improve safety, reduce errors, and ensure compliance with regulatory requirements.
There's three things that control the speed of anything, right?
Time, money and resources.
So the time to adopt cloud technology seems to be perfect.
We are all just coming out of the COVID-19 pandemic, and people started to realize the the real value of the scientific cloud and the value of being able to get your data anywhere.
Money and resources. It's not cheap to do a change management exercise.
It's not cheap to say, “hey, that last 30 years of us buying on-prem software and and various versions of things is going to have to come to a screeching halt and we're gonna have to all go cloud".” That might cost some money and most companies don’t have the right people in place to properly execute that transition.
This is no denigration whatsoever to the amazing scientists, data scientists and scientific engineers who have worked on this for their entire careers.
They've obviously gotten drugs out to market, and have done amazingly well with the tools that they've had to date.
However, you may actually need a more data literate what TetraScience calls “Sciborgs.” People who know both science and tech workflows and can best recommend how scientific data moves from its raw form to its processed and analyzed form two pipelines to destinations.
That's a very rare breed of person, and that kind of person is needed in order to move this ahead and to understand the nuances of large biopharma company workflows. Someone with the know-how to realize they have 23 instruments producing very different data, but they all have to be analyzed in one place — so they suggest maybe putting some commonalities into how those are structured, captured and processed.
While automation can certainly streamline data collection and analysis processes, it is important to recognize that setting up an automated system requires a significant investment of time, resources, and expertise.
Some people may also assume that automated data management systems are "one size fits all," and can be easily applied to all types of data and experiments. In reality, every experiment and data set is unique, which requires a tailored approach to data management and analysis. This means that an effective automation solution must be flexible and adaptable to the specific needs and challenges of each project.
So you can certainly try to build a do-it-yourself (DIY) solution in-house to manage scientific data, but that costs time and money, introduces risk, and shifts focus away from the scientific mission.
But, before you try to go on the journey alone… You should consider a radically better path forward.
The Invisible Assembly Line
Now, let's take a trip back in time to the early days of automobile manufacturing. Henry Ford revolutionized the industry with his assembly line, introducing efficiency and standardization that drastically reduced production time, increased volume, and lowered costs.
But what does this have to do with the biopharmaceutical industry and scientific data automation?
Well, just as the assembly line revolutionized the auto industry, TetraScience is poised to revolutionize the way biopharmaceutical companies approach data management and analysis. We're talking about an invisible assembly line for scientific data workflows, and it's time for biopharma to hop on board.
The biopharmaceutical industry relies on a complex and time-consuming process for developing and producing life-saving drugs. From research and development (R&D) to manufacturing, each step involves specialized expertise, expensive equipment, and regulatory oversight. Not all that dissimilar from the challenges and goals of the automobile industry 100 years ago.
Henry Ford's assembly line revolutionized the manufacturing industry by introducing several benefits that increased efficiency, productivity, and reduced costs:
Reduced production time: Before the assembly line, each worker performed a specific task on a car, and the vehicle was moved from one worker to another. This process was slow and time-consuming, and a car could take several days to complete. With the assembly line, the car moved along a conveyor belt, and each worker performed a small, repetitive task. This reduced production time from several days to just a few hours.
Increased production volume: By reducing production time, the assembly line allowed manufacturers to produce more vehicles in less time. This increased production volume and enabled manufacturers to meet the growing demand for cars.
Lower costs: With the assembly line, each worker became specialized in a specific task, reducing the need for skilled labor. This reduced labor costs and made cars more affordable for consumers. It also allowed Ford to retain key talent as he was able to pay workers $5 a day compared to the 5 cents they previously made, and introduced the concept of the 5 day work week so employees had more time to spend with their families.
Improved quality control: The assembly line introduced a system of quality control that ensured each car met the same standards. Any defects or errors were quickly identified and corrected, reducing waste and improving customer satisfaction.
Standardized parts: With the assembly line, manufacturers standardized parts and components, reducing the need for custom parts and making it easier to replace parts if necessary. This also increased efficiency and reduced costs.
TetraScience is biopharma’s off-the-shelf assembly line for discovery, development, and manufacturing workflows that comes with Sciborg expertise to guide large life science companies through their scientific data journey.
An unexpected and welcomed discovery for many automation engineers, researchers, and scientists at SLAS.
Breathing Life Into Biotech — Funding Announcements
Powered by The Funding Letter
Bicara Therapeutics operates a clinical-stage biotechnology company that engages in the clinical development of bifunctional antibodies that are designed to deliver an immunomodulatory payload directly to the tumor microenvironment, helping cancer patients to ramp up immune cell activity, and potentially offering long-lasting efficacy. View on: LinkedIn.com
New Funding Raised: $108M, Series B
Round Investors: RA Capital Management (lead), Red Tree Venture Capital (lead), Invus, Omega Funds
🚀Growth Focus: Advance its lead program BCA101 and its pipeline of investigational candidates to treat solid tumor cancers.
Press: Business Wire, FinSMEs
HQ: Cambridge, MA
Industry: Biotechnology Research
Employee Count: 16
Noema Pharma is a clinical-stage biotech company that offers novel therapies for conditions affecting the central nervous system including orphan CNS diseases, helping patients with the treatment of neurological and psychiatric indications. View on: LinkedIn.com
New Funding Raised: $112M, Series B
Round Investors: Forbion (lead), Jeito Capital (lead), UPMC Enterprises, Sofinnova Partners
🚀Growth Focus: Support continued development of its diversified pipeline focused on central nervous system disorders.
Press: Business Wire, GlobeNewswire
HQ: Basel, Switzerland
Industry: Biotechnology
Employee Count: 21
Colossal Biosciences is a breakthrough bioscience & genetic engineering company that creates disruptive technologies, and applies CRISPR technology for the purposes of species de-extinction, helping environmental organizations to restore Earth to a healthier state. View on: LinkedIn.com
New Funding Raised: $150M, Series B
Round Investors: US Innovative Technology Fund (lead), Peak 6, Animal Capital, Victor Vescovo
🚀Growth Focus: Advance genetic engineering and pioneer new software, wetware, and hardware solutions, all of which have applications to de-extinction, conservation, and human healthcare.
Press: Business Wire, FinSMEs
HQ: Dallas, TX
Industry: Biotechnology Research
Employee Count: 107 (⚡32% increase in last 6 months)
Lookin’ Around?
Great Jobs in Science & Companies Hiring:
Senior Manager, Research Laboratory — University of Miami, Miller School of Medicine (View Job Post)
Associate, Laboratory Operations — Colossal Biosciences (View Job Post)
Global Head Quality Control (QC) Biologics — Lonza (View Job Post)
Sensing and IoT Senior Research Associate — Corteva (View Job Post)
Chief Data Architect — TetraScience (View Job Post)
Want your company’s job openings posted in future letters for 1300+ life science managers, directors, VPs, and executives to see? Leave the link to your career page in the comments.